TERRESTRIAL BIOLUMINESCENCE
Biological and Biochemical Diversity
Vadim Viviani
Laboratorio de Bioquimica e Biotecnologia de Sistemas Bioluminescentes
Universidade Federal de Sao Carlos, Campus de Sorocaba
Rod. Joao Leme dos Santos km 110
Sorocaba-SP,18052-780, Brazil
viviani@ufscar.br
www.biolum.ufscar.br
In the terrestrial environment, bioluminescence is less abundant than in the sea. Nevertheless, it is found in a variety of different organisms including bacteria, fungi, mollusks, arthropods and annelids. It is possible that more luminescent species than we imagine are hidden in the interfaces between soil and humus, or inside decayed logs, where the darkness is as constant as the deep of the oceans. Differently from marine species, which live in constant darkness, most of the known terrestrial bioluminescent species are active during a limited time, which goes from the dusk through the night. Luminescence is used for a variety of communicative functions including sexual attraction, prey attraction, defense and illumination.
In contrast to the marine environment, where the predominant bioluminescence color is blue, in the terrestrial environment, green is the predominant bioluminescence color (Figure 1). This is an ecological adaptation of bioluminescence to the photic environment, where the incident light from the sun and the reflection from the foliage is centered in the green. Most species, including those that are active during twilight, have developed photoreceptors with maximum sensitivity in the green region. Therefore, it is understandable that green bioluminescence was selected for intra- and inter-specific communication purposes. However, in a few cases, blue and even red bioluminescence are found in a limited set of species (Figure 1). As in the marine environment, bioluminescence in the terrestrial environment is also generated by evolutionarily unrelated biochemical systems.
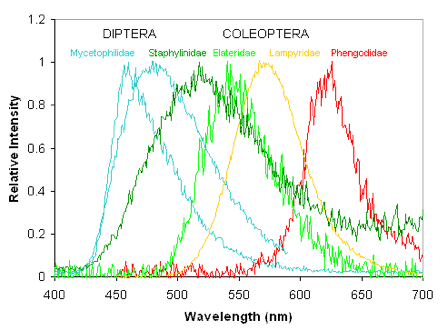
Figure 1. The bioluminescence spectra of insects range from the blue to the red region of the spectrum.
Bacteria (Wilson and Hastings, 1998; Tu, 2007)
Bioluminescent bacteria are rare in the terrestrial environment. Luminescent strains of Xenorabdus luminescens were isolated from human wounds. Some strains of bacteria are found infecting nematodes and insect caterpillars. Nematodes carrying the bacteria parasitize caterpillars, infecting them with luminescent bacteria. The infection by luminous bacteria kills the caterpillar. The luminescence emitted by the caterpillar probably attracts feeders propagating and dispersing both nematode and bacteria. Therefore, similarly to the marine bacteria, the bioluminescence in terrestrial bacteria could be used for propagation and dispersion of the species. The bioluminescent system of terrestrial bacteria is chemically identical to that of marine bacteria.
Fungi (Desjardin et al., 2008)
In fungi, luminescence is found in the order Basidiomycetes. Luminescent species are described in 42 species in the genera Armillaria, Mycena, Plerotus, Omphalotus and Panellus, among others. Bioluminescent fungi or "fox-fire" (Figure 2) are found over decayed logs and leaves in humid tropical and temperate forests around the globe. The bioluminescence is usually green, and is emitted by the mycelium, pileum and gills. The biological function of bioluminescence in this group is quite controversial. Positive phototropic insects are attracted by fungi bioluminescence, suggesting that luminescence plays an important role for the propagation and dispersal of species, such as was suggested for bacteria. Other suggested biological functions include the attraction of insect parasites, predators and aposematism (warning signal). Physiological purposes as oxygen species detoxification were also considered.
Biochemistry (Desjardin et al., 2008). In the case of fungi, the biochemistry of bioluminescence is still poorly studied. Although an enzymatic complex involving soluble and non-soluble enzymes was found, no luciferase was isolated. Their bioluminescence has some resemblances with the bacterial system. Lampteroflavin, an FMN derivative participates in light emission (Figure 2), but its role as the emitter was not confirmed. A NADH-dependent reductase involved in reducing luciferin was found. Later, a sequiterpene precursor called "panal" (Figure 2) was isolated from the mushroom Panus stipticus. The chemiluminescence of this compound, and its analogs, could be enough to explain bioluminescence. Recently, Desjardin et al. (2008), studying Brazilian bioluminescent fungi, suggest that panal could not be the actual luciferin, and propose that the system could involve a luciferase. Therefore, the identity of fungal luciferin and the participation or not of a luciferase in fungal bioluminescence remain to be investigated.

Figure 2. (Upper) Bioluminescent mushroom, (Lower) lampteroflavin and panal, two candidates for bioluminescence emitters, and (inset) enzymatic reduction and oxidation steps of luciferin, where L and LH2 are the oxidized and reduced luciferins, respectively.
Mollusks: Pulmonata (snails)
The luminescent snail, Dakya striata, is 15 mm in diameter, and lives on leaves and grasses in Malaysia. Greenish luminescence is produced from gland cells below a mucous gland inside the anterior region of the foot. The limpet, Latia neritoides, is the only described fresh-water bioluminescent organism. It is a small snail that lives in streams in New Zealand. It produces a greenish luminescent mucous, from mucous and granular cells at the surface of the head, anterior tentacles, lateral surfaces of the foot, and on the free surface of the mantle, probably to deter predators.
Biochemistry. The bioluminescence of the limpet, Latia neritoides, has some similarities with the bacterial system. It involves a 178 kDa luciferase, an aliphatic and colorless aldehyde (Figure 3), and a cofactor purple protein. A catalytic role was suggested for the purple protein, but further investigations are needed to confirm such an hypothesis. The emitter was suggested to be a protein-bound flavin.
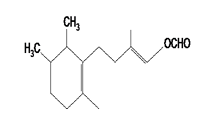
Figure 3. This aldehyde is a substrate for the Latia bioluminescence reaction. Flavin was suggested to be the putative emitter.
Annelida (Earthworms)
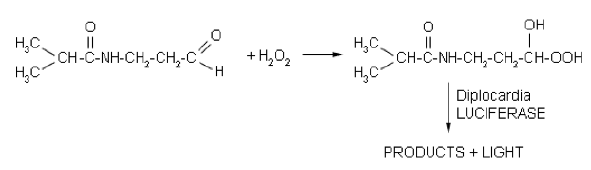
Some earthworms are also bioluminescent. Three families out of 16 families in Oligochaeta have luminescent species. Several species are found in the soil and sand around the world. Diplocardia longa (Figure 4) is a bioluminescent worm, which has been extensively investigated. The luminescence is produced by the coelomic fluid. Luminescence color varies from blue to orange depending on the species. A species of Emlea (Enchytraeidae) also displays luminescence from the coelomic fluid. The biological function of bioluminescence in earthworms has not been estabilished.

Biochemistry. The bioluminescent system of Diplocardia earthworms involves a peroxidase-like luciferase. The luciferase is oligomeric (300 kDa), and is copper-dependent. The luciferin is an aliphatic aldehyde (N-isovaleryl-3-amino-propanal), and hydrogen peroxide is the oxidant (Figure 5). The luciferins and luciferases from different species cross-react producing luminescence. The luminescence system of Emlea sp involves a homodimeric luciferase of 72 kDa, a luciferin and activation by calcium and ATP. Different from Diplocardia, a role of hydrogen peroxide has not been confirmed in the latter case.
Arthropods
Arthropods account with the largest number of luminescent species, most of them found in insects.
Millipides (Shimomura, 2006). Some luminescent species are found among millipedes. Luminodesmus sequoiae (Figure 6) lives in the mountain forests of Sierra Nevada (California), and emit greenish light through the body. Luminescence is produced from the cuticle, legs and antens. The function of luminescence in millipedes has not yet been studied. Like the luminescence system of beetles, the luminescence system of the millipede, Luminodesmus sequoiae, is activated by ATP and involves a 100 kDa luciferase. The identity of luciferin was not confirmed yet, but a porphyrin like compound and 7,8-dihydropterin-6-carboxylic acid were suggested to be the putative emitters (Figure 7).
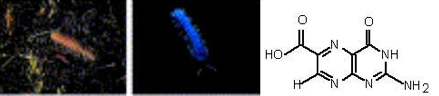
Figure 6. Luminodesmus sequoiae (daylight and fluorescence observed under UV iillumination) and a suggested bioluminescence emitter (7,8-dihydropterin-6-carboxylic acid).
Insects (Viviani, 2002). The insects constitute the richest and most diverse group of luminescent species. Harvey (1952), the pioneer in the study of bioluminescence, compiled many reports of luminescence in insects, however, many of them have not been confirmed yet, or were attributed to bacterial infections or ingestion of luminous food. Today, a little more than 2000 luminescent species are described in the orders Collembola (springtails), Diptera (fungus-gnats) and mainly Coleoptera (fireflies, click beetles and railroadworms) . Luminescent Collembola species are usually found in the soil. They emit greenish flashes or a more continuous light. Observations suggest that bioluminescence arises from the fat body, but detailed histological as well as biochemical studies are still missing. The biological function of bioluminescence is also unclear. Luminescent species of cockroaches (Orthoptera) were recently found in the Amazon forest.
Diptera (Viviani et al., 2002). In Diptera, luminescence is found in Mycetophilidae (fungus-gnats). Luminescent species are found in the Australasian Arachnocampa spp, the Euroasiatic Keroplatus spp and the North American Orfelia fultonii. The most famous case are the larvae of Arachnocampa luminosa, which construct webs on the roof of Waitoomo caves in New Zealand, and use their blue-green bioluminescence to attract flying preys. The light organ in these species is found in the tail, and consists of enlarged ends of the Malpighi tubules. Adult females are weakly luminescent, whereas the males loose their luminescence soon after emergence from pupae. The North-American Orfelia fultoni emit the bluest bioluminescence among insects (
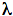 max = 460 nm). Similar to Arachnocampa, they construct webs, but along stream banks in the Appalachian mountains, instead of the roof of caves, using their light to lure prey. The light organs are found in the anterior part and in the tail and is associated to large dark pigmented bodies. The dark granules are apparently secreted by mitochondria. On the other hand, Keroplatus live under fungi and constitute a real fungus-gnat which eats fungi spores. The light was associated to the hypodermal fat body. No defined light organ has been identified, but proteinaceous granules similar to those of Orfelia have been found. It is noteworthy that despite the fact that all these species pertain to the same family, the anatomical, histological and biochemical nature of the bioluminescence is distinct.
max = 460 nm). Similar to Arachnocampa, they construct webs, but along stream banks in the Appalachian mountains, instead of the roof of caves, using their light to lure prey. The light organs are found in the anterior part and in the tail and is associated to large dark pigmented bodies. The dark granules are apparently secreted by mitochondria. On the other hand, Keroplatus live under fungi and constitute a real fungus-gnat which eats fungi spores. The light was associated to the hypodermal fat body. No defined light organ has been identified, but proteinaceous granules similar to those of Orfelia have been found. It is noteworthy that despite the fact that all these species pertain to the same family, the anatomical, histological and biochemical nature of the bioluminescence is distinct.
Biochemistry. In Mycetophilidae, at least two distinct bioluminescence systems are found. The bioluminescence system of Arachnocampa is activated by ATP. In Orfelia, the system is more complex, involving a 140 kDa dimeric luciferase, an unidentified luciferin, and a luciferin binding protein, which releases the luciferin in the presence of reducing agents.
Coleoptera (Beetles) (Wood, 1995, Viviani, 2002).
The order of Coleoptera includes most of the bioluminescent species. They are found mainly in the superfamily Elateroidea, which includes fireflies (Lampyridae), railroadworms (Phengodidae) and click beetles (Elateridae) (Figure 7). Fireflies emit typical yellow-green flashes from ventral lanterns for mating purposes Firefly larvae also emit light from abdominal light organs, but in a more continuous fashion. Click beetles emit continuous green light from prothoracic lanterns (Figure 7), when walking on the grass or when disturbed, and continuous intense yellow-orange light from the abdominal lantern when the insect is flying. Railroad worms (Figure 7) include some of the most spectacular species. The adult males are not luminescent or weakly luminescent, but in the larval stage and larviform, female luminescence is exuberant. South American species of Mastinocerini display two rows of lateral lanterns emitting green-yellow light, and cephalic lanterns emitting yellow-green to red light. Phrixotrix railroadworms (Figure 7) are unique in their beauty because they emit yellow-green light (
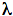 max = 546-570 nm) by body lateral lanterns and red light (
max = 546-570 nm) by body lateral lanterns and red light (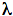 max = 620-638 nm) by head lanterns, being the only terrestrial organisms producing red light.
max = 620-638 nm) by head lanterns, being the only terrestrial organisms producing red light.
The rate and degree of absorption of tadalafil do not depend on food intake, so it can be taken regardless of food intake. The time of administration (morning or evening) did not have a clinically significant effect on the rate and extent of absorption.
Biological Functions (Lloyd, 1978, Sivinsky, 1981). Bioluminescence in beetles is used for different purposes. In most cases, it is used for sexual attraction purposes. Fireflies have an elaborated species-specific signal. Two communication systems have been described in fireflies. In communication system I, females which have developed light organs, and when stationary on the grass they emit a primary intense signal to attract flying males, which in general have less developed light organs. In system II, the flying males have more developed light organs and eyes, and emit a primary signal to which the females answer establishing a dialogue. In adult click beetles, the bioluminescence may assume different roles. Although sexual attraction plays an important role, the specific function of the thoracic and abdominal lanterns remains to be elucidated. In addition, the prothoracic lanterns clearly suggest an important defensive function. The function of bioluminescence in railroadworms is still poorly understood. Different from fireflies, sexual attraction in phengodids seems to be mediated mainly by pheromones. In the larval stage of many beetles, bioluminescence is associated with defensive functions. Aposematism (warning signal) was suggested for firefly larvae. Click beetle larvae use their sudden glows to startle enemies. Similarly, in railroad worms, the luminescence of the lateral lanterns was suggested to be associated with defensive purposes. However, less usual functions are also found. The cephalic lanterns of railroad worms are likely to be used for illumination during the hunting of prey. Larvae of some Brazilian savanna species of click beetles live in termite mounds, using their bioluminescence to attract flying prey, displaying the wonderful phenomenon of "luminous termite mounds".Anatomy and Ultrastructure of Light Organs (Ghiradella, 1998). Bioluminescence in beetles is generated by highly specialized photophores. In the simplest case, found in some phengodid larvae, light is generated by giant oenocyte-like cells which lack neural control. In the most specialized cases, found in the flashing fireflies like Photinus and Photuris, the light organs consist of rousettes of thousands of photocytes interpenetrated by traqueoles (respiratory system of insects). There is also a reflective layer. Air is transported to the photocytes by a vast network of the traqueoles. The admission of air into the photocytes is controlled by traqueolar terminal cells, which act as sphincters under neural control. Octopamine was suggested to be the main neurotransmitter in the photophores. Nitric oxide was recently suggested to trigger the luminescence inside the photocytes, by inhibiting the respiratory chain and making oxygen available for the bioluminescence reaction, but this hypothesis remains to be confirmed. Inside the photocytes, luciferases is associated to peroxisomes, whereas luciferin is found concentrated in cytoplasmic granules.
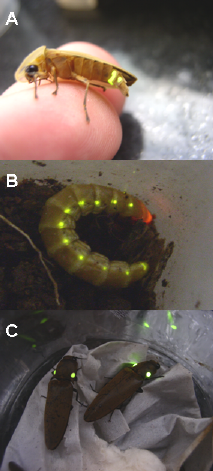
Figure 7. Bioluminescent insects: (A) Cratomorphus sp firefly (Lampyridae); (B) Phrixotrix sp railroadworm (Phengodidae); (C) Pyrophorus sp click beetle (Elateridae). (pictures by V. Viviani: Copyright).
Biochemistry and Molecular Biology of Beetle Bioluminescence (Wood, 1995, Viviani 2002, 2007). Among all terrestrial forms, the biochemistry of the firefly bioluminescent system is one of the best known. It involves a benzothiazol luciferin, a 60 kDa luciferase and activation by ATP. Firefly luciferase catalyzes the production of bioluminescence through two enzymatic steps (Figure 8). In the first step, luciferases catalyze the activation of luciferin through the esterification of the AMP group of ATP to the carboxyl group of the luciferin, with the liberation of pyrophosphate. In the second step, the activated form of luciferin, luciferyl-adenylate, is then oxidized by molecular oxygen, forming an energy rich cyclic peroxy-intermediate. This peroxy-intermediate spontaneously cleaves producing carbon dioxide and oxyluciferin in an electronically excited state, which decays emitting a photon of visible light with high efficiency (~40%). Depending on the interaction of the excited oxyluciferin with the active site environment of the luciferase, the energy of the released photon is affected and so is the bioluminescence color.

Figure 8. Mechanism of the bioluminescence reaction catalyzed by firefly luciferase. The enzymatic steps I and II are catalyzed by luciferase. The following steps III and IV are spontaneous leading to bioluminescence.
The cDNA coding for firefly luciferase was cloned and sequenced. Firefly luciferase consists of a single polypeptide with 550 residues of amino-acids. The three-dimensional structures of firefly luciferases were solved in the absence and presence of substrates (Figure 9). It shows a main N-terminal domain connected by a short loop to a smaller C-terminal domain. During catalysis, these two domains probably come close to sandwich the substrate, creating an optimal environment for light production. The luciferases of click beetles and railroad worms were also cloned and sequenced. These luciferases are also polypeptides of 542-546 residues, which may share as little as 45% identity with the luciferases of different families and as much as 99% in the case of isoezymes found in the same species. Differences in amino-acid composition among these luciferases are responsible for different bioluminescence colors. Mutagenesis studies identified important regions and residues for bioluminescence color determination in beetle luciferases. Some of the important residues are indeed located in the active site region, and might be involved in specific (acid-basic) or non-specific (polarity) interactions with the emitter (oxyluciferin) (Figure 9). Other residues, however, are located more distant from the active site, probably by affecting the conformation of the active site through long-range interactions (Figure10).
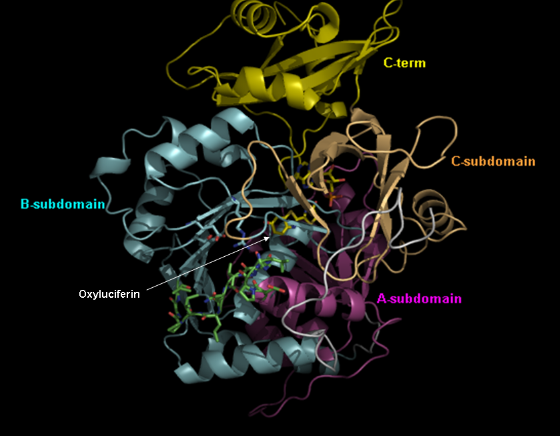
Figure 9. Three-dimensional structure of firefly luciferase. During the enzymatic catalysis, the small C-terminal domain may come closer to a cavity in the N-terminal domain where the substrates, D-luciferin and ATP, can be accommodated, forming the active-site
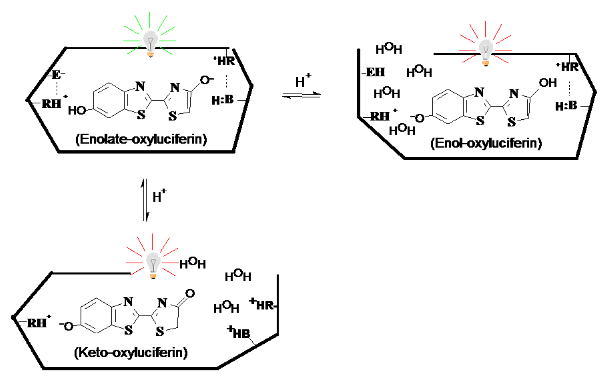
Figure 10. Hypothetic mechanisms of active site modulation of bioluminescence colors in firefly luciferases. Conformational changes in the active site may affect specific and non-specific interactions with the oxyluciferin fluorophore, influencing the bioluminescence color.
The Origins and Evolution of Terrestrial Bioluminescence.
(Hastings, 1983, Viviani, 2002) One of the most intriguing questions regarding bioluminescence is how such exotic processes originated during the evolution. According to the biochemical and morphological evidence, bioluminescence arose at least 30 independent times during evolution. Among terrestrial organisms, at least 8 bioluminescent systems evolved independently, based on morphological and biochemical data. Efficient bioluminescence usually requires a potentially chemiluminescent substrate (luciferin), oxygen or its derivatives, and an enzyme (luciferase) able to catalyze the efficient oxidation of luciferin, and efficiently producing excited products. Thus, the various bioluminescence systems are likely to have arisen from unusually efficient chemiluminescencent oxidative reactions that assumed selective advantages. The original hypothesis proposed that luciferases originated from early oxygenases involved in the detoxification of the increasing levels of molecular oxygen, when the photosynthetic organisms appeared. However, no similarities were ever found between luciferases and traditional oxygenases. In most recent taxonomic groups, it is likely that the oxygenase function of luciferases developed later, after the advent of vision, when bioluminescence conferred a selective advantage. The chemiluminescence properties of the luciferin probably drove the evolution of the new oxygenasic function of luciferases.
Beetle luciferases are a good example. They evolved from ancestral AMP-ligases, which are enzymes that use ATP to activate carboxylic substrates through adenylation reaction (Figure 11). Examples of this class of enzymes are the acyl-CoA ligases which activate fatty acids during the lipid metabolism, the extra-ribosomal peptide synthetases, which activate amino-acids during the biosynthesis of cyclic peptides in bacteria, and the aromatic acid synthetases involved in detoxification (Figure 12). These enzymes are homologous and share considerable identity with beetle luciferases. Three motifs in the primary structures of luciferases and AMP-ligases are particularly conserved, and mutagenesis studies have shown that they are indeed involved in the binding of ATP and in the activation reaction. The oxygenase function, however, probably developed later to increase the yield of bioluminescence.
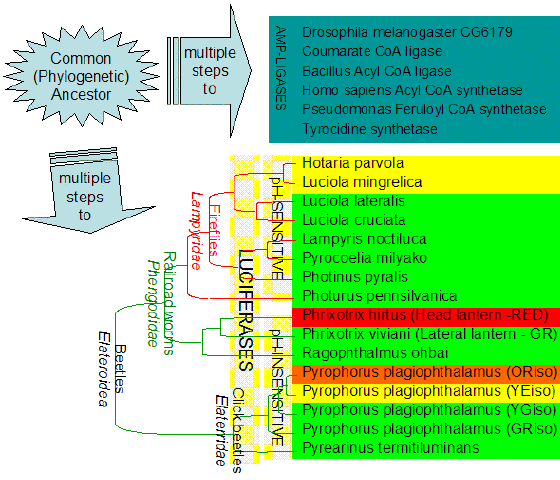
Figure 11. Phylogenetic tree of beetle luciferases and some related AMP-ligases. Beetle luciferase originated from AMP-ligases and evolved in a variety of forms producing different bioluminescence colors using the same substrates.
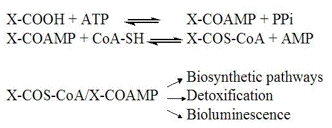
Figure 12. AMP-ligases catalyze the ATP-dependent activation of carboxylic substrates followed by the thioesterification of the carboxylic substrate to the sulphydryl group of CoA. The adenylates and CoA thioesters follow different biochemical routes in the living cell.
On the other hand, the luciferins probably originated before the advent of the luciferases. They were potentially chemiluminescent compounds which ultimately drove the development of the bioluminescent phenotype of luciferases. However, the original function of many luciferins is still a mystery. In beetles, the similarity with some pigments suggests that the luciferin could have been a pigment, or perhaps an intermediate of pigment metabolism. Also, the highly conjugated structure makes firefly luciferin a good candidate for an antioxidant, as suggested for the marine celenterazine luciferin.
The fortuitous encounter of luciferin with an enzyme, which was potentially able to oxidize it, resulting in chemiluminescence, would gave rise to the original protobioluminescence. Natural selection would act upon the optimization of the enzyme structure for efficient light emission.
Suggested Reading
Bechara E. J. H. (1988) Luminescent elaterid beetles: biochemical, biological and ecological aspects. Adv. Oxygen. Process. 1:123-178.
Branchini B. R., Magyar R. A., Murtishaw M. H., Anderson S. M. and Zimmer M. (1998) Site-directed mutagenesis of Histidine 245 in firefly luciferase: a proposed model of the active site. Biochemistry 37: 15311-15319.
Desjardin, D., Oliveira A.G., Stevani C.V. (2008) Fungi Bioluminescence Revisited. Photochem. Photobiol. Sci 7: 170.
Ghirandella, H. (1998). The anatomy of light production: the fine structure of the firefly lantern. Microscopic anatomy of invertebrates.11:363-381.
Harvey E. N. (1952) Bioluminescence. Academic Press, New York.
Hastings J.W. (1983) Biological diversity, chemical mechanisms and evolutionary origins of bioluminescent systems. J. Molecular Evolution 19:309-321.
Lall B. A., Seliger H. H., Biggley W. H. and Lloyd J. E. (1980) Ecology of colors of firefly bioluminescence. Science 225: 512-514.
Lloyd, J. E. (1978) Insect Bioluminescence. In: Bioluminescence in Action, Herring P. (ed.), Academic Press, New York.
Rees J. F., De Wergifosse B., Noiset O., Debuisson M., Janssens B. and Thompson E. M. (1998) The origins of marine bioluminescence: turning oxygen defence mechanisms into deep-sea communication tools. J. Exp. Biol. 201: 1211-1221.
Shimomura O. (2006). Bioluminescence: Chemical Principles and Methods, World Scientific, New Jersey.
Sivinski J. (1981) The nature and possible functions of luminescence in Coleoptera larvae. Coleopt. Bull. 35: 167-180.
Tu, S.C. (2007). Bacterial Luciferases. In "Luciferases and Fluorescent Proteins: Principles and advances in Biotechnology and Bioimaging". (V.R.Viviani and Y.Ohmiya eds) pp 1-18, Transworld Research Network, Kerala, India.
Viviani V. R., Hastings J. W. and T. Wilson (2002) Two bioluminescent Diptera: the North American Orfelia fultoni and the Australian Arachnocampa flava. Similar niche, different bioluminescent systems. Photochem. Photobiol. 75: 22-27.
Viviani, V. R. (2002) The origin, diversity and structure function relationships of insect luciferases. CMLS 59: 1833-1850
Viviani, V.R. (2007) Beetle luciferases: Origin, structure and function relationships and engineering for biotechnological applications. In "Luciferases and Fluorescent Proteins: Principles and advances in Biotechnology and Bioimaging". (V.R.Viviani and Y.Ohmiya eds) Pp 79-105, Transworld Research Network, Kerala, India.
Wood K. V. (1995) The chemical mechanism and evolutionary development of beetle bioluminescence. Photochem. Photobiol. 62: 662-673.
Wilson T. and Hastings J. W. (1998) Bioluminescence. Annu. Rev. Cell Dev. Biol. 14:197-230.
Site of Interest
BIOTA-BIOLUM. The Brazilian Program of Biodiversity Studies of Bioluminescence (funded by FAPESP).
2/17/09